TB 500
- For in vitro testing and laboratory use only.
- Not for human or animal consumption.
- Bodily introduction is illegal.
- Handle only by licensed professionals.
- Not a drug, food, or cosmetic.
- Educational use only.
Researchers in the early 2000s noticed something unusual about the protein thymosin beta-4: it was the most abundant intracellular protein in mammalian cells, found in nearly every tissue type, and it appeared to play a central role in cellular migration. Wherever cells needed to move — into wounds, toward damaged tissue, into developing organs — thymosin beta-4 was somehow involved. The 2004 Bock-Marquette paper in Nature established that Tβ4 activates integrin-linked kinase and promotes cardiac cell migration, survival, and repair — and a 2007 Smart et al. paper in Nature followed, demonstrating it could also mobilize epicardial progenitor cells and drive neovascularization in the adult heart after injury. Together, those two studies made TB-500 famous as a regenerative research compound.
What Is TB-500?
This is where the confusion starts. "TB-500" is sold commercially in two distinct forms, and they're often labeled identically:
The full molecule: Synthetic thymosin beta-4 (Tβ4) — a 43-amino-acid peptide identical to the natural protein, sometimes with N-terminal acetylation for stability. This is the version with the bulk of published research behind it. The 2004 Bock-Marquette and 2007 Smart Nature papers, the 2003 Philp diabetic wound-healing study, and most clinical trial work used full-length Tβ4.
The fragment: A 7-amino-acid heptapeptide (sequence Ac-LKKTETQ, residues 17-23 of full Tβ4), also called Fequesetide. This is the actin-binding domain of Tβ4 isolated as a standalone fragment. Smaller, cheaper to synthesize, but with substantially less direct research validation than the full molecule.
Both forms appear in the research peptide market under the name "TB-500." When you buy TB-500, you need to know which one you're getting — they have different molecular weights (4,963 Da for full Tβ4 vs 889 Da for the LKKTETQ fragment), different research bases, and different pharmacological profiles.
The Actin-Binding Mechanism That Defines Both Forms
What both versions share: they bind G-actin (globular actin) monomers, sequestering them and preventing premature polymerization into F-actin (filamentous actin). That's the foundational mechanism. Cells need actin polymerization to move, change shape, and remodel their environment — and they need precise control over when polymerization happens. Tβ4 is one of the master regulators of that timing.
The downstream effects flow from actin regulation. Cell migration. Endothelial movement (angiogenesis — new blood vessel formation). Wound closure. Inflammatory cell recruitment. Cardiac progenitor cell mobilization. The 2003 Philp study (Wound Repair and Regeneration) demonstrated 2-3 fold stimulation of keratinocyte migration at concentrations as low as 10 picograms. The 2004 Bock-Marquette Nature paper established cardiac cell migration, survival, and ILK activation. The 2007 Smart et al. Nature paper extended the work to epicardial progenitor cell mobilization and neovascularization. Multiple clinical trials investigated Tβ4 for ischemic stroke, traumatic brain injury, and corneal injury — most ended without progressing to FDA approval.
What Serious Buyers Should Know
Here's the uncomfortable truth: most clinical research on Tβ4 has been disappointing. The compound has been investigated for over two decades. Phase 2 trials in stroke, traumatic brain injury, and venous stasis ulcers ended without breakthrough efficacy. RegeneRx Biopharmaceuticals, the company that holds most of the original Tβ4 patents, has struggled to advance any indication to FDA approval despite consistent preclinical signals. The mechanism is real. The animal data is consistent. The translation to human therapeutic outcomes has been substantially less impressive than the early research suggested.
For research applications — wound healing models, cardiac regeneration studies, actin biology, angiogenesis research — the compound remains a useful tool with decades of mechanistic literature behind it. For drawing conclusions about specific human therapeutic outcomes, the gap between preclinical promise and clinical evidence is substantial.
Regulatory note: Thymosin Beta-4 (full-length) was placed on the FDA's Category 2 bulks list in 2023. On April 22, 2026, the FDA removed Thymosin Beta-4 from Category 2 after the original nominations were withdrawn. PCAC review is scheduled for July 23, 2026 alongside BPC-157, KPV, and MOTS-c. As of May 2026, TB-500 sits in regulatory transition — no longer formally restricted, not yet formally approved for compounding. WADA prohibits TB-500 for athletes in tested sports under category S2.
Why Generic Peptides for TB-500?
Here's a sourcing problem that's specific to TB-500: the full Tβ4 vs fragment confusion is endemic in the research peptide market. Cheap suppliers routinely sell the 7-amino-acid LKKTETQ fragment under the "TB-500" label at full Tβ4 prices, or mix the two in inconsistent ratios. The two compounds have completely different research bases — most published Tβ4 research used the full 43-amino-acid molecule, not the fragment. If you're trying to replicate published Tβ4 cardiac regeneration or wound healing data, accidentally getting the heptapeptide fragment means your experiment isn't what you think it is. The full molecule is also significantly more expensive and technically demanding to synthesize correctly, which creates strong incentives for cheap producers to substitute the cheaper fragment.
Generic Peptides supplies research-grade TB-500 for sale at 99% purity, manufactured in the USA. Domestic synthesis with verified molecular identity — the part that determines whether you have full-length Thymosin Beta-4 or the LKKTETQ fragment, and whether your research data matches the published literature accordingly.
Order TB-500 for sale in the USA — 99% purity, full Thymosin Beta-4 sequence verified, manufactured domestically.
TB-500 FAQ
Is it legal to buy TB-500 in the US for research?
Yes — TB-500 is legally available as a research compound in the United States. As of April 22, 2026, Thymosin Beta-4 has been removed from FDA Category 2, with PCAC review scheduled for July 23, 2026 to determine its compounding status. It's not FDA-approved for human use. WADA prohibits it for tested athletes.
Is TB-500 the same as Thymosin Beta-4?
Not always — and the confusion is endemic in the market. "TB-500" can refer to either full-length synthetic Thymosin Beta-4 (43 amino acids) or the LKKTETQ heptapeptide fragment (7 amino acids, residues 17-23). Most published research used the full 43-amino-acid molecule. Cheap suppliers often sell the fragment under the TB-500 label at full Tβ4 prices.
What does the published research actually show?
Animal data is consistent and impressive across multiple tissue types — wound healing, cardiac regeneration, endothelial migration, angiogenesis. Human clinical translation has been substantially less successful. RegeneRx Biopharmaceuticals' Phase 2 trials in stroke, TBI, and venous stasis ulcers ended without breakthrough efficacy. The mechanism is real; the magnitude of human therapeutic effect has been disappointing.
What's the difference between TB-500 and BPC-157?
Different mechanisms, often used together in research. BPC-157 (a 15-amino-acid pentadecapeptide) works primarily through angiogenesis and growth factor signaling at local injury sites. TB-500 (Tβ4) works through G-actin sequestration affecting cell motility and migration. Researchers studying recovery often examine both individually before combining them.
I've seen TB-500 sold cheap online — same product?
Probably not the same compound. Cheap suppliers routinely sell the 7-amino-acid LKKTETQ fragment as "TB-500" while charging full Tβ4 prices, or deliver mixed/inconsistent product. Without analytical verification of which molecule is actually in the vial, the price gap often reflects that you're paying full price for a structurally different (and substantially cheaper) compound.
Sources
Bock-Marquette I et al. — "Thymosin beta4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair." Nature, 2004. Foundational research on Tβ4 cardiac cell migration, survival, and ILK activation. https://pubmed.ncbi.nlm.nih.gov/15565145/
Smart N et al. — "Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization." Nature, 2007. Documents epicardial progenitor cell activation and cardiac repair after injury. https://pubmed.ncbi.nlm.nih.gov/17108969/
Philp D, Badamchian M, Scheremeta B et al. — "Thymosin beta 4 and a synthetic peptide containing its actin-binding domain promote dermal wound repair in db/db diabetic mice and in aged mice." Wound Repair and Regeneration, 2003. Documents wound healing in impaired models. https://pubmed.ncbi.nlm.nih.gov/12581422/
Goldstein AL, Hannappel E, Kleinman HK — "Thymosin beta4: actin-sequestering protein moonlights to repair injured tissues." Trends in Molecular Medicine, 2005. Comprehensive review of Tβ4 mechanism and wound healing applications. https://pubmed.ncbi.nlm.nih.gov/15953756/
FDA — "Bulk Drug Substances Nominated for Use in Compounding Under Section 503A," updated April 22, 2026. Documents Thymosin Beta-4 / TB-500 removal from Category 2 and the July 23, 2026 PCAC consultation. https://www.fda.gov/media/94155/download
Two molecules sold under one name. Identity verification determines what you're actually researching.
TB-500 Storage Guide: How to Keep Your Research Peptide Stable and Effective
TB-500 (Thymosin Beta-4) ships as a white lyophilized powder in a sealed glass vial, freeze-dried to preserve this 43-amino-acid peptide and extend its shelf life. With a few simple habits — cold, dark, dry — the sealed vial stays in perfect condition for its full shelf life. Here's exactly how to store it.
Lyophilized Powder (Unreconstituted)
| Parameter | Details | Notes |
|---|---|---|
| Storage Temperature | Freezer at −20°C (−4°F) for long-term storage up to 24 months. Refrigeration at 2–8°C (36–46°F) is fine for short-term use up to ~3 months. | Original sealed vial in the freezer is the safest default. |
| Light Sensitivity | Yes — protect from direct light and UV exposure to prevent photodegradation. | Keep in the original box or an opaque, amber container. |
| Freezing | Allowed and recommended. −20°C is standard for long-term storage; −80°C extends stability further if available. | Freeze from the start if you won't use it within 3 months. |
| Oxidation Sensitivity | TB-500 contains a methionine residue that's prone to oxidation if the vial seal is broken or the powder is exposed to air. | Keep the aluminum crimp cap intact until ready to reconstitute. |
| Signs of Degradation | Healthy powder is white to off-white and loose or cake-like. Watch for yellowing, browning, clumping, visible moisture, or a sticky texture. | Any color change, clumping, or moisture = discard the vial. |
| Common Mistakes | Leaving the vial at room temperature after delivery, storing in a humid kitchen or bathroom, or opening a cold vial and letting condensation form inside. | Put it in the freezer on arrival, and let sealed vials warm to room temperature before opening. |
Shipping & Product Authenticity
Every order is processed quickly and shipped with full tracking. All products come directly from the official Generic Peptides supply chain — in original manufacturer packaging, carefully handled from warehouse to your door.
Shipping Times
| Destination | Delivery Time | Notes |
|---|---|---|
| USA Domestic | 2–5 business days | Faster when local warehouse stock is selected at checkout |
| International | 10–15 business days | Tracking included; update frequency may vary by destination country |
| Order Processing | 24–48 business hours | Processing begins after payment confirmation |
| Tracking | Provided on all orders | Tracking number sent after dispatch; multiple warehouses may result in separate shipments |
Direct Supply & Secure Delivery
This product is supplied through the official Generic Peptides distribution chain and shipped in original manufacturer packaging. Orders are packed securely to protect the contents during transit and to respect customer privacy as a standard practice.
Outer packaging is neutral and does not display product details on the exterior — a common approach to protect shipments from damage, tampering, and unnecessary exposure during delivery.
What to Expect
- Orders are processed after payment confirmation
- USA domestic shipping is typically faster when local stock is selected
- International orders include tracking, though update frequency may vary by destination
- Multiple warehouses may result in separate shipments when applicable
Authenticity & Verified Supply
Every order includes full authenticity assurance: official Generic Peptides presentation, batch-linked lab documentation, and sealed original packaging — giving customers confidence in every purchase.
| Authenticity Feature | Details |
|---|---|
| Packaging | Original manufacturer packaging — sealed and unaltered |
| Lab Documentation | Batch-linked certificate of analysis available on request |
| Supply Chain | Sourced exclusively through official Generic Peptides distribution |
Shipping & Returns
Certificate of Analysis (COA)
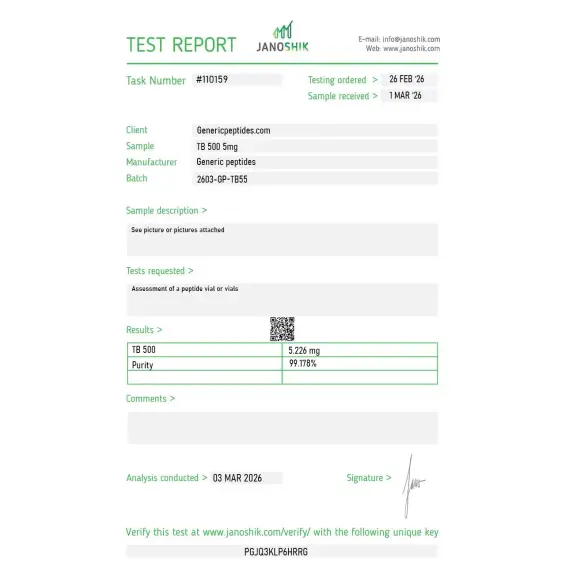
Independent lab test reports are available for TB-500 2mg, 5mg, and 10mg batches. Each certificate provides batch-level verification details, including measured content, purity result, analysis date, and report documentation.



Actin-sequestering assays in endothelial cells. Behaved as the literature predicts.
Goldstein isolated this from calf thymus back in 1981 and it's been studied as a defined protein ever since — refreshing to work with something in this catalog that has a real, multi-group literature behind it rather than a single research program. Material gave clean results in our angiogenesis tube-formation assays.
RegeneRx ran actual Phase 2 and Phase 3 trials on this — RGN-259 for ophthalmic, the cardiac and dermal programs. That's a genuine clinical evidence base, more than almost anything else in this space. We used it for corneal epithelial migration work and the material tracked with the published behavior.
Reorder for angiogenesis work. Full Tβ4, consistent.
We run Tβ4 against BPC-157 in parallel regenerative models — different mechanisms entirely, actin-sequestering versus whatever BPC does, but they're the two compounds everyone studies side by side in this area. Getting both from one source with matched documentation keeps the comparison honest. That's the value for this kind of work.
Endothelial migration assays. No issues, three orders.
Solid for our cardiac-repair preclinical models, where the angiogenic signaling is what we're after. The 43-mer is a moderately large peptide and I'd push the vendor to include mass-spec intactness verification as standard — longer sequences carry more truncation-product risk than the small peptides, and "purity by HPLC" alone doesn't fully reassure me at this length. Material checked out when I ran it myself.
The product page actually frames this as actin-cytoskeleton biology rather than a "recovery peptide," which is the correct scientific framing and almost nobody uses it. Tβ4's primary intracellular role is G-actin sequestering — the regenerative effects are downstream and extracellular. Nice to see a listing that leads with the real biology.
For corneal wound-healing research the RGN-259 development trajectory makes Tβ4 the obvious reference, and the material's been consistent enough to run a multi-timepoint migration study without re-validating between batches. The fact that there's a real ophthalmic clinical program behind it gives the in-vitro work a translational anchor.
Reliable in our scratch-wound migration assays. Reconstituted cleanly. The one ongoing friction is that Tβ4 aggregates somewhat in solution at higher concentrations, so we aliquot and keep working stocks dilute — not a vendor issue, just the peptide's behavior, but worth others knowing before they prepare concentrated stocks and watch them cloud.
TB-500 (Thymosin Beta-4) binds G-actin (globular actin) monomers through a high-affinity binding interface, sequestering them and preventing polymerization into F-actin (filamentous actin) until cellular signals trigger release. This actin-sequestering activity regulates the timing and location of cytoskeletal reorganization required for cell migration, wound healing, and tissue remodeling. Downstream effects include endothelial cell movement, keratinocyte migration, and progenitor cell mobilization.
Length and research base. Full Tβ4 is a 43-amino-acid endogenous protein found throughout mammalian cells. The TB-500 fragment (Ac-LKKTETQ) is a synthetic 7-amino-acid peptide derived from residues 17-23 of full Tβ4 — its actin-binding domain isolated as a standalone fragment. Most published Tβ4 research used the full molecule, not the fragment.
Different mechanisms targeting overlapping research applications. BPC-157 works primarily through angiogenesis and local growth factor signaling. TB-500 works through G-actin sequestration affecting cell motility and migration. Researchers studying tissue repair often investigate both individually before combining them. The two compounds have largely independent research literatures despite the popular co-mention.
Two main issues. First, the full-vs-fragment confusion: cheap suppliers routinely sell the 7-amino-acid LKKTETQ fragment as "TB-500" while charging full Tβ4 prices. Second, the full 43-amino-acid Tβ4 has hydrophobic regions and aggregation tendencies that make synthesis technically demanding — manufacturing yields typically range 30-50% after purification, and cheap producers cut quality to manage costs.
Thymosin Beta-4 was first isolated from thymus tissue in the 1970s as one of the original "thymosin" peptides. Synthetic full-length Tβ4 entered active research in the 1990s and 2000s. The TB-500 designation emerged from the research peptide and veterinary medicine markets, with the 2007 Bock-Marquette Nature paper marking a major expansion of cardiac research applications.
Yes. WADA prohibits TB-500 (Thymosin Beta-4) for athletes subject to drug testing under category S2 (Peptide Hormones, Growth Factors, Related Substances and Mimetics). The classification covers all forms — full Tβ4, the LKKTETQ fragment, and related analogs. Athletes in tested sports should avoid the compound regardless of which form is used or how it's obtained.
Thymosin Beta-4, Tβ4, TB4, Timbetasin (the proposed INN for full Tβ4), Fequesetide (for the LKKTETQ fragment), and various commercial designations. Full Tβ4 has CAS number 77591-33-4. The LKKTETQ fragment has CAS number 476014-70-7. Different naming conventions across literature reflecting whether the focus is the full molecule or the fragment.
Wound healing and tissue regeneration research lead by volume — keratinocyte migration, dermal wound repair, and corneal injury models. Cardiac regeneration research expanded substantially after the 2007 Bock-Marquette work. There's also active research in angiogenesis, anti-inflammatory mechanisms, hair follicle biology, and neurological injury models including stroke and traumatic brain injury.
RegeneRx Biopharmaceuticals advanced Tβ4 through multiple Phase 2 trials in stroke, traumatic brain injury, and venous stasis ulcers without achieving breakthrough efficacy that justified Phase 3 progression. The pattern — strong preclinical signals followed by disappointing human translation — is common with regenerative compounds. The mechanism appears robust at the cellular level; the magnitude of clinical benefit has been less impressive than animal data suggested.
Most regenerative peptides target specific signaling pathways (growth factors, angiogenic mediators) or tissue-specific receptors. TB-500 operates through actin cytoskeletal regulation — a fundamental cellular machinery that affects how cells move and reshape themselves. That foundational mechanism is what gives Tβ4 its breadth of effects across tissue types and is also what makes its action difficult to isolate from other cellular processes.
Researchers investigating tissue repair, cellular migration, and actin-dependent regenerative biology consistently examine TB-500 alongside compounds that target overlapping or complementary recovery and repair pathways. BPC-157 is the defining pairing in the research literature — TB-500 works through G-actin sequestration affecting cell motility and migration while BPC-157 works through angiogenesis and growth factor signaling at local injury sites; the two mechanisms are complementary enough that researchers studying musculoskeletal recovery almost always examine both individually before combining, and the TB-500/BPC-157 stack is the most-studied peptide combination in tissue repair research. IGF-1 LR3 provides systemic anabolic and tissue-repair signaling through IGF-1R activation — a hormonal approach to the same recovery outcomes that TB-500 addresses through cytoskeletal biology, making it useful for dissecting mechanical vs hormonal contributions to muscle regeneration. PEG-MGF targets satellite cell activation specifically — TB-500's cell migration effects and PEG-MGF's satellite cell proliferation effects address complementary aspects of the muscle regeneration sequence, and researchers studying the full cellular cascade from damage through repair often examine both. Thymosin Alpha-1 occasionally appears in the same research context given TB-500's origin as a thymic peptide — both are derived from thymus tissue and both affect immune cell function, and researchers studying the immune component of tissue repair sometimes examine both simultaneously.